Neuromuscular ultrasound in leprosy: A scanning protocol
Renann Pirola MD(1) | Leo H. Visser MD, PhD(2) | Yasmin K. Nasr-Eldin MD(3) | Monika Krzesniak-Swinarska MD(4) | Stephen Wilkes Wheat MD(5) | Michael S. Cartwright MD, MS(6)
(1)Department of Neurology, HUCAM, Vitciria, Brazil. (2) Department of Neurology and Clinical Neurophysiology, ETI Hospital, Tilburg, The Netherlands. (3)Rheumatology and Rehabilitation Department. Minia University, Al Minya, Egypt. (4)Department of Neurology, University of New Mexico, HSC, Albuquerque, New Mexico. (5)Department of Neurology-Guest Lecturer. Baylor College of Medicine. Houston. Texas. USA. (6)Department of Neurology, Wake Forest School of Medicine. Winston-Salem, North Carolina. USA. Correspondence Renann Pirola, Department of Neurology. HUCAM, Rua Saul Navarro, 200, Ap. 303, Vitciria-ES 29055-360, Brazil. Email: dr.pirolaneuro@gmail.com
Abstract: The diagnosis of leprosy neuropathies has been traditionally based on clinical findings and electrodiagnostic studies, but ultrasound has emerged as a new tool for use in clinical practice. We conducted a literature search on the subject and developed a pragmatic ultrasound scanning protocol for patients with confirmed or suspected leprosy neuropathy. We suggest scanning the ulnar, median, superficial radial, common fibular and sural nerves at specific sites and assessing cross-sectional area, vascularity, and epineural thickness. Our protocol is potentially useful in differentiating leprosy neuropathies from other demyelinating neuropathies, but its applicability and accuracy must be evaluated in different centers.
KEYWORDS: demyelinating. leprosy, mononeuropathy, neuropathy, ultrasound
1 | INTRODUCTION
Leprosy is a chronic bacterial infection caused by Mycobacterium /eprae, a pathogen transmitted by inhalation and with tropism for phagocytes in the skin and 5chwann cells within the peripheral nerves.(1)1 It most commonly involves the skin with a wide variety of cutaneous lesions, but peripheral nerves are the second most affected site.(2) The incubation period ranges from 3 months to 40 years, with an average of 7 years(3) and symptoms vary between two poles accordingly to the host immune response. Initially, patients often present with hypopigmented ill-defined macules, usually with thermal sensory impairment over the lesions. This stage is known as indeterminate leprosy and it can evolve to either one of the poles. Patients with a strong cellular immunity usually develop erythematous macules with well-defined borders known as tuberculoid leprosy while those with a weak cellular immunity usually develop multiple macules and papules that progress to diffuse skin infiltration. This is known as lepromatous leprosy and it can affect other sites like upper respiratory tract, lymph nodes, and joints.
Classifications based on clinical manifestations reflect this disease spectrum and serve mainly for research purposes. The Ridley-Jopling classification has tuberculoid (TT) and lepromatous (LL) leprosy as opposite ends of this spectrum which also includes borderline tuberculoid (BTI. borderline borderline (BB) and borderline lepromatous (BL) stages. World Health Organization (WHO) developed a pragmatic classification based on the number of cutaneous lesions and on the presence of acid-fast bacilli (AFB) in slit-skin smears (555). Patients are classified as having paucibacillary leprosy when they have 5 or fewer lesions and no AFB in 555 and as having multibacillary leprosy when they have 6 or more lesions or AFB present in 555.
The peripheral nerve involvement starts in superficial dermal sensory branches and causes areas of hypoesthesia or anesthesia. Initially, it predominates in small fibers related to thermal and pain sensation, but all sensory modalities are affected later on. As the disease progresses, it tends to affect larger nerve trunks, with large-fiber sensory and motor symptoms. There is a predilection for cooler and more superficial areas of the body and the ulnar nerve is by far the most commonly affected nerve.
Three clinical features of leprosy neuropathies must be emphasized: asymmetry, clinical course, and the absence of pain. Leprosy neuropathies are typically asymmetric and present as a mononeuropa· thy or multiple mononeuropathies. and even when a pattern of polyneuropathy is found, it is most commonly from confluent involvement of multiple nerves. The mononeuropathies are usually slow progressing, painless, and easily distinguished from more acutely presenting asymmetrical inflammatory neuropathies. Nonetheless, it is important to have in mind that cases of leprosy reactions can present acutely or subacutely and with prominent pain. Nerve damage in tuberculoid leprosy can be considered more focal and there is involvement of nerves that surround the cutaneous lesions, usually with enlargement and marked asymmetry. Lepromatous leprosy neuropathy usually shows a more diffuse pattern, with more confluent abnormalities.
Nerve involvement has traditionally been diagnosed based on clinical examination and electrodiagnostic studies, but nerve ultrasound has emerged as a new sensitive and painless tool that supports the diagnosis.(4·5) We aimed to review the current literature and create a protocol for ultrasound scanning of individuals suspected of having leprosy with peripheral nerve involvement based on clinical signs and symptoms.
2 | METHODS
Our first step was to review the current literature on the topic. A search was performed on PubMed, Medline and Cochrane databases with the words "leprosy" and "ultrasound (searching up to August 21, 2023) and duplicates, articles without abstract and written in any other language than English were excluded. We screened each publication to assess if it really provided information on the use of nerve ultrasound to diagnosis or follow-up patients with suspected or confirmed leprosy neuropathies and particular attention was paid to which nerves were assessed, what segments of these nerves were assessed and what were the abnormalities described in each one of the articles. We had 56 final results in our search: 26 analytical studies published in indexed journals, 10 analytical studies presented as con· ference abstracts, 14 case reports or case series and 5 narrative review.
The first author was in charge of the literature review and sent a summary of the articles to the other authors along with the full articles and commentaries on what each study assessed and what were their outcomes. After the review. the first author created a scanning protocol considering when to use nerve ultrasound in leprosy, the nerves most commonly involved, the most common sites of involvement and the most important abnormalities described in the literature. This first draft was then sent to all the authors and there was the first round of discussion when each one gave their opinion on what should be added or excluded in the protocol and a general discussion about all the comments that were made during this first round. After this first round of discussion, the first and last authors decided what to change in the proposed protocol based on the comments made by all the authors, on how these comments were supported by the literature and on how it would impact on feasibility. The first author created the second protocol based on the implemented changes and there was then the second round of discussion when the first and second authors explained what was changed in the first protocol and the reasons for these changes and the other authors were once more able to make additional comments. After this second round of discussion, the first and last authors considered all the comments and developed this final version of the protocol.
3 | RESULTS
3.1 | When to use nerve ultrasound?
We considered four different clinical scenarios in which nerve ultrasound may be helpful:
1. Patients with diagnosed cutaneous leprosy and possible neural involvement.
2. Patients treated for leprosy with subsequent worsening or recurrent nerve symptoms.
3. Patients with suspected pure neural leprosy (PNL).
4. Patients with idiopathic neuropathies in regions endemic for leprosy.
The first scenario includes patients with dermatological manifestations of leprosy who may or may not have neural symptoms, and some studies found in the literature were done in these patients aiming to identify nerve involvement.(5•11) Ultrasound scanning can be used to confirm the suspected involvement of a specific nerve, to better assess patients with "confluent"' involvement, and also to screen for subclinical involvement in patients without neural symptoms. Sonographic assessment has a higher diagnostic yield than clinical evaluation and electrodiagnosis alone when it comes to detecting abnormalities of leprosy neuropathies(7·12) and provides additional ana· tomical information.
The second scenario encompasses patients with worsening or recurrent nerve symptoms despite treatment. Leprosy reactions are inflammatory responses that may occur before, during or after treatment and carry additional risk of nerve damage. Since its treatment is based on different types of immunotherapy, early recognition is important and many studies were done with this aim.(4·12·13) Ultrasound imaging also offers the possibility of following and comparing nerve abnormalities over time, and despite some authors discussed about that.(13·14)
In the third scenario, we proposed, ultrasound could help in suspected cases of PNL. It is a form of the disease without skin involve· ment and with neural symptoms only, in which diagnostic confirmation usually relies on nerve biopsy. Ultrasound can be useful to identify which nerves are involved and the sites of involvement, to suggest biopsy sites, and to support the diagnosis when biopsy is not an option. There is one original article which assessed clinical, neurophysiological, sonographic and pathological features of 100 patients with PNL and used ultrasound to include patients in the probable degree of diagnostic certainty.(15)
The fourth scenario is in patients with idiopathic neuropathies, a challenge in clinical practice. It is difficult to select whom among those patients deserve further investigation with invasive procedures such as nerve biopsy or more expensive tests such as genetic testing. In regions endemic for leprosy, an isolated ulnar neuropathy may raise suspicion about this diagnosis, but it is not easy to decide when to refer the patient for dermatological evaluation or proceed with invasive investigation. In these scenarios, we think ultrasonographic findings may help in the decision. In the literature, most of the studies were done with leprosy patients and healthy volunteers and studies which directly compare the sonographic pattern of leprosy neuropathies to other types of neuropathies are still lacking.
3.2 | What nerves should be imaged?
Given the typical clinical and sonographic asymmetry observed in patients with leprosy,(9) we recommend bilateral scanning even if symptoms are only unilateral, as this approach will allow comparisons between symptomatic and asymptomatic sides.
When deciding which nerves to scan, it is important to take into consideration the most commonly affected nerves. The ulnar and common fibular nerves are the most commonly affected nerves in leprosy, with 88% and 34% of involvement, respectively,(16) and it has been suggested that the cooler temperatures of these regions favor bacterial proliferation,(17) although this is not confirmed. The posterior tibial nerve is involved in 20% of the cases,(18) and the median nerve may be involved less often, with variables rates in the literature. Other nerves often involved are the superficial radial, dorsal ulnar cutaneous, great auricular, supraorbital branch of the trigeminal nerve, and sural nerve.(19) Bilateral scanning of all these nerves would be timeconsuming and likely not informative for every patient. Therefore, our recommended approach is based on the previous considerations as well as the feasibility of the protocol.
For patients who are in one of the four clinical scenarios outlined above, it is important to consider if the patient has any suspected nerve involvement based on history, examination, and electrodiagnostic testing (if performed). If the symptoms raise suspicion of a mononeuropathy, then scanning the suspected nerve is the first recommended step. Otherwise, if the symptoms are more diffuse and show a pattern of multiple mononeuropathies or polyneuropathy, or if the patient does not have any neural symptoms, then scanning all the nerves would be time-consuming. For these cases, we suggest bilateral scanning of ulnar, median and superficial radial nerves as a first step. If no abnormalities are found in these nerves, or if there is a high clinical suspicion for involvement of nerves of the lower limbs, then we suggest proceeding with the scanning of bilateral common fibular and sural nerves.
Our decision of including the ulnar nerve is supported by the literature, since it is the nerve most commonly involved both clinically and sonographically.(5·10•13·15) Although the median nerve is not as commonly enlarged as the ulnar,(15) we decided to include it in our protocol because of the important role that the assessment of this nerve has in other acquired demyelinating neuropathies like chronic inflammatory demyelinating polyneuropathy(20) and how this can possibly be further investigated in future studies. Although only one study assessed the superficial radial nerve in leprosy patients,(15) we decided to scan it in our protocol to include a pure sensory nerve in the upper limbs which can be chosen a site of biopsy. We decided for the superficial radial nerve over the dorsal ulnar cutaneous nerve because the latter would already been seen branching from the main ulnar nerve scanned in the protocol.
We decided to put the nerves in the lower limbs as a second step mostly because of the aimed feasibility of our protocol, since scanning these nerves is more technically demanding and time consuming. We decided to include the common peroneal nerve because it is the nerve most commonly involved in the lower limbs(15) and the sural nerve because it is the one classically chosen as a site of biopsy.
3.3 | How to scan the nerves of interest?
We will discuss how to scan the nerves based on the most common sites of involvement in leprosy.
We suggest the following scanning sites:
• Ulnar nerve -> from the mid-forearm to the axilla
• Median nerve -> from the wrist to the axilla
• Superficial radial -> from the distal forearm to the mid-foreann
• Common fibular nerve -> from the popliteal fossa to just below the fibular head
• Surat nerve -> from the ankle to the mid-calf
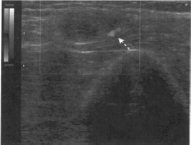
The ulnar nerve can be scanned by starting at the mid-forearm where it courses under the flexor carpi ulnaris (FCU) and over the flexor digitorum profundus (FOP), and then by moving the transducer proximally while following the nerve in cross-section (Figure 1). It is useful to have the patient with the elbow slightly flexed and with the shoulder slightly abducted to better expose the medial elbow. The nerve passes between the two heads of the FCU and underneath the ligament connecting these two heads (Osborne's fascia) and courses proximally posterior to the medial epicondyle. The ulnar nerve is usually affected slightly proximal to the elbow, peaking at four centimeters above the ulnar sulcus,(21•22) so we suggest scanning at least six centimeters proximal to the medial epicondyle, and at times scanning to the axilla based on symptoms and suspicion of undetected ulnar involvement.
The median nerve can be scanned from the wrist to the axilla (Figure 2). At the wrist, the nerve is superficial within the carpal tunnel and it dives deep in the forearm while coursing proximally between the flexor digitorum superficialis and the FOP. In the proximal forearm it courses between the two heads of the pronator teres and in the distal arm it is medial to the brachia! artery. The median nerve can be traced up to the axilla, where it is located lateral and superior to axillary artery. It is important to emphasize that carpal tunnel syndrome is a common disorder and that median nerve enlargement at the wrist in a leprosy patient may not necessarily be related to leprosy. Just like the ulnar nerve, leprosy median nerve enlargement tends to be more proximal than in carpal tunnel syndrome.(23)
The superficial radial nerve can be easily found at the distal onethird of the lateral forearm where it has a subcutaneous course superficial to the radius; starting from this point the transducer is then moved proximally (Figure 3). A single measurement of this nerve approximately 9 cm proximal to the distal radius is recommended. Another option is to start scanning the radial nerve at the elbow, with the transducer in the groove between the brachialis and the brachioradialis. The nerve can be found superficial to the brachialis and deep to the brachioradialis and can be followed distally. The radial nerve usually bifurcates into its superficial and deep branch at the elbow, with a classical "snake eyes" appearance. Since it is a purely sensory branch, the superficial radial nerve is often considered as a possible site of biopsy when affected.
The common fibular nerve can be scanned by starting at the popliteal fossa (Figure 4). With the transducer at the popliteal fossa, it can be moved proximally and distally looking for the bifurcation of the sciatic nerve into the fibular and tibial nerves. The fibular nerve is the most lateral branch of the bifurcation and can be followed distally while it courses around the fibular neck, its common entrapment point. Sometimes it may be challenging to find the nerve at the popliteal fossa and starting at the fibular neck might be higher yield.
The sural nerve is a small sensory branch. It is usually found between the lateral malleolus and the Achilles tendon, accompanied by the lesser saphenous vein (Figure 5), and it can be followed proximally in the calf. Another option would be to start scanning proximally, finding the nerve between the two heads of the gastrocnemius and following it distally to the ankle. The sural nerve is usually the site of choice for nerve biopsy when affected.
It is helpful to compare the nerve findings in leprosy patients to those in the general population, and normal reference values of cross-sectional areas (CSAs) have been published for the main sites of each nerve.(24)








3.4 | What measurements and assessments are informative?
When scanning a peripheral nerve, we can analyze many of its sanegraphic parameters. Some of them are: CSA on axial view, thickness on longitudinal view, echogenicity, fascicular architecture, vascularity, mobility, and epineural thickness.
CSA on axial view is the cornerstone measurement for nerve ultrasound. It is the most studied parameter in the literature and there are CSA reference values for the main nerves. When measuring the CSA of a nerve, we must place the marker just inside the hyperechoic border of the epineurium so that a comparison can be made to a set of normal reference values (Figure 6). Thickness measurements on longitudinal view are also useful, but they are more prone to error because identifying the thickest portion of the nerve in longitudinal view can be challenging. CSA is the parameter most commonly assessed in the literature reviewed. (5•8)
The sonographic appearance of a normal nerve on axial view is that of a "honeycomb," with hypoechoic fascicles surrounded by the hyperechoic perineurium, all within the hyperechoic epineurium. With injuries, nerves tend to become hypoechoic and lose fascicular structure.
Vascularity within the nerve can be assessed with either color Doppler (CD) or power Doppler (PD) modes (Figure 7). The normal nerve usually does not show positive Doppler signal with these modes, but increased vascularity has been described in leprosy neuropathies, especially in the context of acute reactions.(12)
Epineural thickness is a parameter analyzed less often than CSA, but there is evidence of increased epineural thickness in leprosy patients.25 Reference values for epineural thickness in leprosy and healthy patients have been published for the ulnar nerve (Figure 8).(25)
We suggest scanning the nerves and identifying the site of maximum enlargement. Measurements of the CSA and epineural thickness and assessment of vascularity should then be performed at the enlargement sites and compared to published reference values or to the contralateral side (if asymptomatic).
4 | DISCUSSION
To our knowledge, this is the first sonographic scanning protocol for leprosy neuropathies developed from a review of the scientific literature. Although it is based on the current literature, decisions were made by balancing feasibility and appropriate evaluations for a disease which is pleomorphic and typically multifocal.
Checking the applicability and accuracy of our protocol in different centers will bring further information about its usefulness. If proved applicable and accurate, our protocol can be used to confirm clinical involvement of nerves, to detect subclinical involvement and to follow-up patients during and after treatment.
As our knowledge of sonographic patterns of other neuropathy etiologies grow, comparing these patterns to the pattern in leprosy patients by using our protocol may give us practical information for decision-making in practice. This may be particularly useful in endemic regions where leprosy is part of the differential diagnosis of demyelinating neuropathies, especially when access to nerve biopsy is difficult or impossible.
Since we included pure sensory nerves, in centers where nerve biopsy is available, our protocol can be used in addition to clinical examination and electrodiagnostic studies to select the best site for biopsy. Given the multifocal nerve involvement in these patients, ultrasound has the advantage of assessing which part of the nerve in enlarged, potentially increasing the diagnostic yield of the biopsy.
There are limitations to the approach we used in generating this article. Firstly, it is based on a review of the literature and expert opinion, which can be informative but it is also prone to biases. Secondly, the literature on ultrasound in leprosy is not extensive, so there were not a lot of large studies to guide our approach. Thirdly, our approach has not been tested for diagnostic accuracy in a systematic, prospective study. Finally, our approach has not been tested to determine if it ultimately improves patient outcomes in a cost-effective manner.
5 | CONCLUSION
This article outlines a pragmatic approach to the use of nerve ultrasound for the diagnostic assessment of individuals with leprosy and/or suspected leprosy and nerve involvement. Future, large, prospective, and multicenter trials testing our approach for diagnostic accuracy and improvement of patient outcomes is recommended as the next step for assessing these protocols.
CONFLICT OF INTEREST STATEMENT - The authors declare no conflicts of interest.
DATA AVAILABILITY STATEMENT - Data sharing is not applicable to this article as no new data were created or analyzed in this study.
ETHICS STATEMENT - We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
PATIENT CONSENT STATEMENT - Verbal consent.
ORCID - Renann Pirola https://orcid.org/0000-0003-2926-570X
REFERENCES
1. Chen KH, Lin CY, Su SB, Chen KT. Leprosy: a review of epidemiology, clinical diagnosis, and management. J Trop Med. 2022;2022:8652062. doi:10.1155/2022/8652062
2. Lastoria JC, Abreu MA. Leprosy: review of the epidemiological, clinical, and etiopathogenic aspects -part 1. An Bras Dermatol. 2014; 89(2):205-218. doi:10.1590/abd1806-4841.20142450
3. Ooi WW, Srinivasan J. Leprosy and the peripheral nervous system: basic and clinical aspects. Muscle Nerve. 2004;30(4):393-409. doi:10. 1002/mus.20113
4. Jain S, Visser LH, Praveen TL, et al. High-resolution sonography: a new technique to detect nerve damage in leprosy. PloS Neg/ Trap Dis. 2009;3(8):e498. doi:10.1371/journal.pntd.0000498
5. Venugopal R, Binesh VG, Puthussery PV, George S, Asokan N. Comparison of high resolution ultrasonography with clinical examination in the assessment of peripheral nerve involvement in leprosy. Indian. Dermatal Online J. 2021;12(4):536-540. doi:10.4103/idoj.lDOJ_ 720_20
6. Spitz CN, Mogami R, Pitta UR, et al. Ultrasonography as a diagnostic tool for neural pain in leprosy. PLoS Neg/ Trap Dis. 2022;16(4): e0010393. doi:10.1371/journal.pntd.0010393
7. Sreejith K, Sasidharanpillai S, Ajithkumar K, et al. High-resolution ultrasound in the assessment of peripheral nerves in leprosy: a comparative cross-sectional study. Indian J Dermatol Venereal Leprol. 2021;87(2):199-206. doi:10.25259 /IJDVL_ 106_20
8. Gupta S, Bhatt S, Bhargava SK, Singal A. Bhargava S. High resolution sonographic examination: a newer technique to study ulnar nerve neuropathy in leprosy. Lepr Rev. 2016;87(4):464-675.
9. lugao HB, Nogueira-Barbosa MH, Marques W, Foss NT, Frade MA Asymmetric nerve enlargement: a characteristic of leprosy neuropa· thy demonstrated by ultrasonography. PLoS Neg/ Trap Dis. 2015;9(12): e0004276. doi:10.1371/journal.pntd.0004276
10. Frade MA, Nogueira-Barbosa MH, Lugao HB, Furini RB, Marques Junior W, Foss NT. New sonographic measures of peripheral nerves: a tool for the diagnosis of peripheral nerve involvement in leprosy. Mem Inst Oswaldo Cruz. 2013;108(3):257-262. doi:10.1590/S0074- 02762013000300001
11. Kumaran MS, Thapa M, Narang T, Prakash M, Dogra S. Ultrasonography versus clinical examination in detecting leprosy neuropathy. Leprosy Rev. 2019;90(4):364-370.
12. Akita J, Miller LHG, Mello FMC, et al. Comparison between nerve conduction study and high-resolution ultrasonography with color doppler in type 1 and type 2 leprosy reactions. Clin Neurophysiol Pract. 2021;17(6):97-102. doi:10.1016/j.cnp.2021.02.003
13. Chaduvula MV, Visser LH, Suneetha S, et al. High-resolution sonography as an additional diagnostic and prognostic tool to monitor disease activity in leprosy: a two-year prospective study. Ultraschall Med. 2018;39(1):80-89.
14. Lugao HB, Frade MA, Marques W Jr, Foss NT, NogueiraBarbosa M H. Ultrasonography of leprosy neuropathy: a longitudinal prospective study. PLoS Negl Trap Dis. 2016;10(11):e0005111. doi:10. 1371/journal.pntd.OOOS111
15. Shukla B, Verma R, Kumar V, et al. Pathological, ultrasonographic, and electrophysiological characterization of clinically diagnosed cases of pure neuritic leprosy. J Peripher Nerv Syst. 2020;25(2):191-203. doi: 10.1111/jns.12372
16. Yadav N, Kar S, Madke B, et al. leprosy elimination: a myth busted. J Neurosci Rural Pract. 2014;S(suppl 1):S28-S32. doi:10.4103/0976- 3147.145197
17. Vijayan J, Wilder-Smith EP. Neurological manifestations of leprosy, chapter 2.5. In: Scollard DM. Gillis TP, eds. International Textbook of Leprosy. American Leprosy Missions; 2016. www.internationaltext bookofleprosy.org
18. Vijayan BV, Dominic MR, Nair VCP. Leprous neuropathy: observational study highlighting the role of electrophysiology in early diagnosis. J Neurosci Rural Pract. 2021;12(3):530-534. doi:10.1055/ s-0041-172757S
19. van Brake! WH, Nicholls PG, Wilder-Smith EP, et al. Early diagnosis of neuropathy in leprosy -comparing diagnostic tests in a large prospective study (the INFIR cohort study). PLoS Neg/ Trap Dis. 2008;2(4): e212. doi:10.1371/journal.pntd.0000212
20. Yun CJ, Crump N, Puckett M, Cartwright MS. Focused neuromuscular ultrasound approach for the diagnosis of chronic inflammatory demyelinating polyneuropathy. J Clin Neurophysiol. 2023;40(4):378-381. doi:10.1097 /WNP.0000000000000905
21. Bathala L, Krishnam VN, Kumar HK, et al. Extensive sonographic ulnar nerve enlargement above the medial epicondyle is a characteristic sign in Hansen's neuropathy. PLoS Neg/ Trap Dis. 2017;11(7): e0005766. doi:10.1371/joumal.pntd.0005766
22. Bathala L, Kumar K, Pathapati R, Jain S, Visser LH. Ulnar neuropathy in Hansen disease: clinical, high-resolution ultrasound and electrophysiologic correlations. J Clin Neurophysiol. 2012;29(2):190-193. doi: 10.1097/WNP.0b013e31824d969c
23. Nagappa M, Pujar GS, Keshavan AH, et al. Sonographic pattern of median nerve enlargement in Hansen's neuropathy. Acta Neural Scand. 2021;144(2):155-160. doi:10.1111/ane.13432
24. Cartwright MS, Passmore LV, Yoon JS, Brown ME, Caress JB, Walker FO. Cross-sectional area reference values for nerve ultrasonography. Muscle Nerve. 2008;37(5):566-571. doi:10.1002/mus.21009
25. Visser LH, Jain S, Lokesh B, Suneetha S, Subbanna J. Morphological changes of the epineurium in leprosy: a new finding detected by high-resolution sonography. Muscle Nerve. 2012;46(1):38-41. doi:10. 1002/mus.23269
How to cite this article: Pirola R, Visser LH, Nasr-Eldin YK, Krzesniak-Swinarska M, Wheat SW, Cartwright MS.
Neuromuscular ultrasound in leprosy: A scanning protocol. J Clin Ultrasound. 2023;1-7. doi:10.1002/jcu.23591

